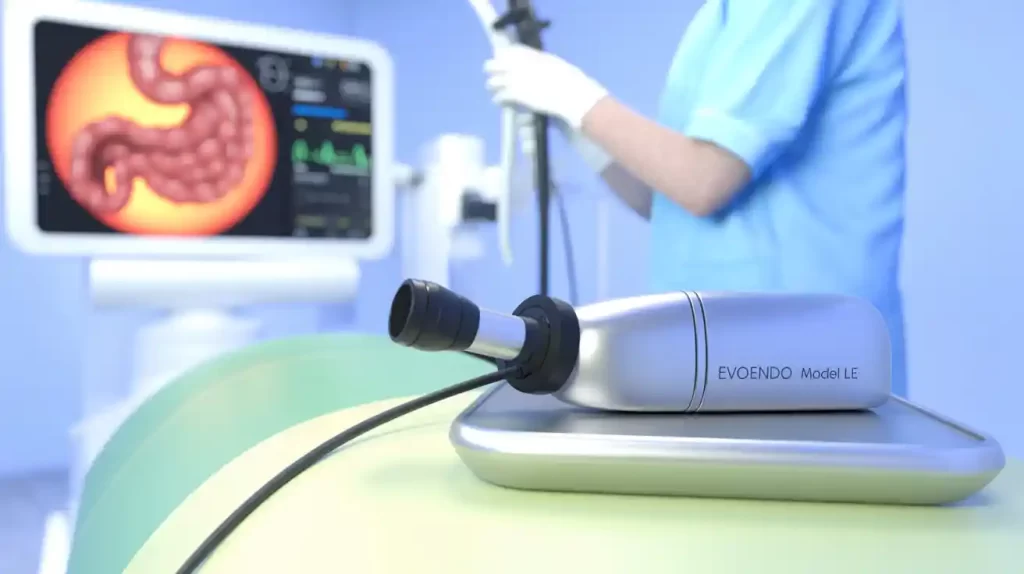
EvoEndo has received expanded FDA clearance for its Model LE gastroscope, now approved for patients of all ages—from neonates to adults.
Previously cleared for ages five and up, the updated indication makes EvoEndo the only provider of transoral endoscopy for infants using ultra-slim scopes. The company already led in sedation-free, transnasal endoscopy for older pediatric patients.
With ChannelMax technology supporting standard pediatric accessories and scope lengths of 85 and 110 cm, EvoEndo’s system is designed to address a wide range of anatomies and procedures. Its portable platform also enhances care accessibility across diverse clinical settings.
CEO Jonathan Hartmann emphasized the milestone as “elevating care for newborns and infants” while broadening endoscopy access across age groups.
Follow MEDWIRE.AI for pediatric MedTech breakthroughs.