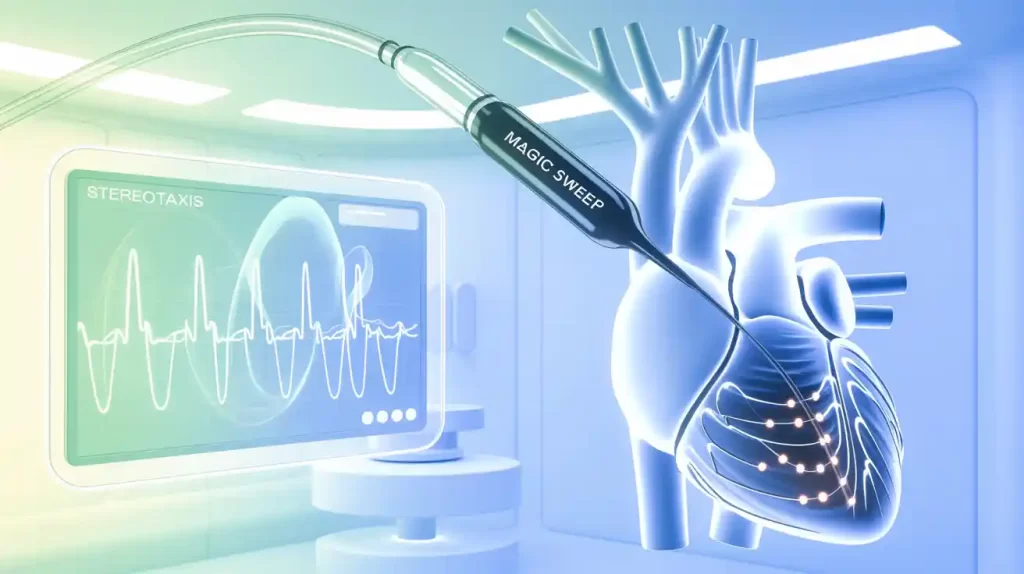
Stereotaxis (NYSE: STXS) received FDA 510(k) clearance for its Magic Sweep navigation catheter, the world’s first robotically navigated high-density electrophysiology (EP) mapping catheter. Featuring 20 electrodes, Magic Sweep delivers rapid, detailed electroanatomical heart chamber maps to improve the diagnosis and treatment of complex arrhythmias.
Designed with an atraumatic shaft, the catheter avoids tissue distension common with rigid catheters, enabling safer, more accurate mapping. Magic Sweep integrates with Stereotaxis’ robotic systems, enhancing precision and workflow as the company develops automated mapping algorithms.
“This FDA clearance marks a pivotal moment for Stereotaxis,” said CEO David Fischel, highlighting plans for multiple upcoming robotically-steered devices.
The company plans a broad commercial launch of Magic Sweep in 2025.
Follow MEDWIRE.AI for updates on robotic innovations in electrophysiology.