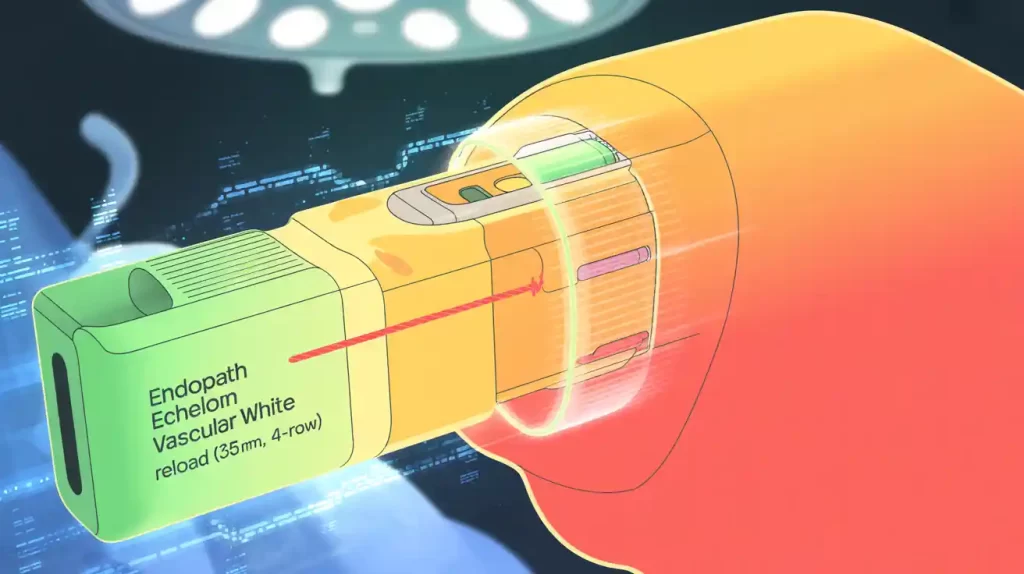
Johnson & Johnson MedTech’s Ethicon Endo-Surgery has issued a device correction for its Endopath Echelon vascular white reload cartridges used with the Echelon Flex Powered Vascular Stapler. The FDA flagged this as a serious recall, involving an inadvertent lockout during surgery where the device momentarily activates but fails to cut or staple tissue.
Though the device lockout does not cut or staple, it requires additional steps to safely release and remove it, posing a risk of serious injury or death. Ethicon has reported one death and one injury related to this issue. The design includes safeguards to prevent vessel injury, with staples remaining below the tissue surface and the knife not advancing fully during lockout.
Ethicon advises users to review updated instructions and provides a video demonstration on managing device lockouts. The correction affects specific product lots of the Endopath Echelon Vascular White Reload (35mm, 4-row) with product code VASECR35.
Follow MEDWIRE.AI for surgical device safety updates.