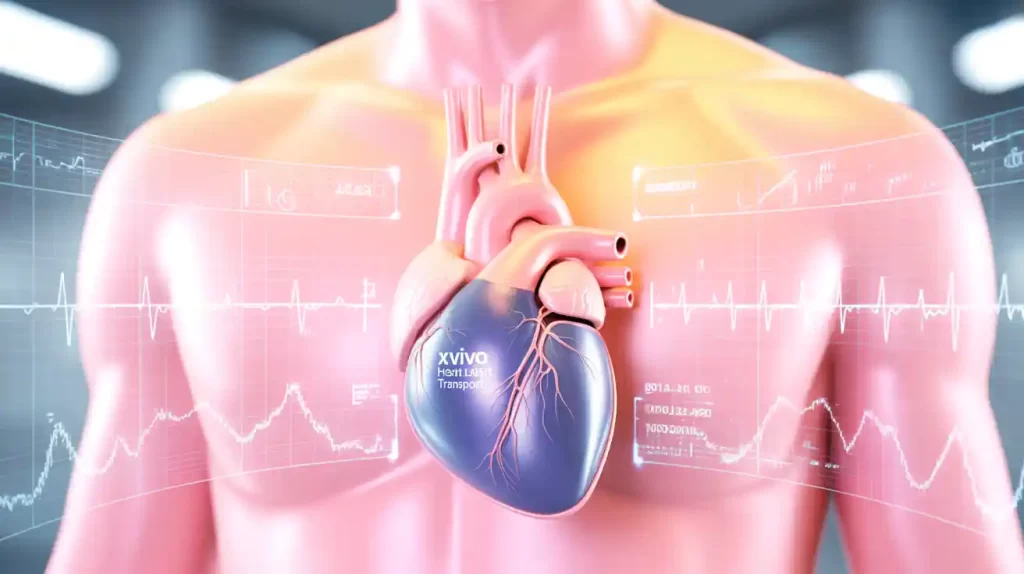
Xvivo announced the enrollment of the first patient in its continued access protocol (CAP) study for the Heart Assist Transport device. The study follows the PRESERVE trial in the U.S. and allows up to 60 patients to be enrolled across 26 transplant centers.
The FDA-approved CAP enables ongoing clinical use of the device while collecting one-year follow-up data in preparation for a premarket approval (PMA) submission. The protocol closely mirrors the original investigational device exemption (IDE) study and will remain active until FDA completes the PMA review or enrollment is complete.
CEO Christoffer Rosenblad emphasized the study as a key step toward expanding access to life-saving technology and supporting clinicians in advanced heart care.
Follow MEDWIRE.AI for latest updates on transplant and cardiac device innovations.