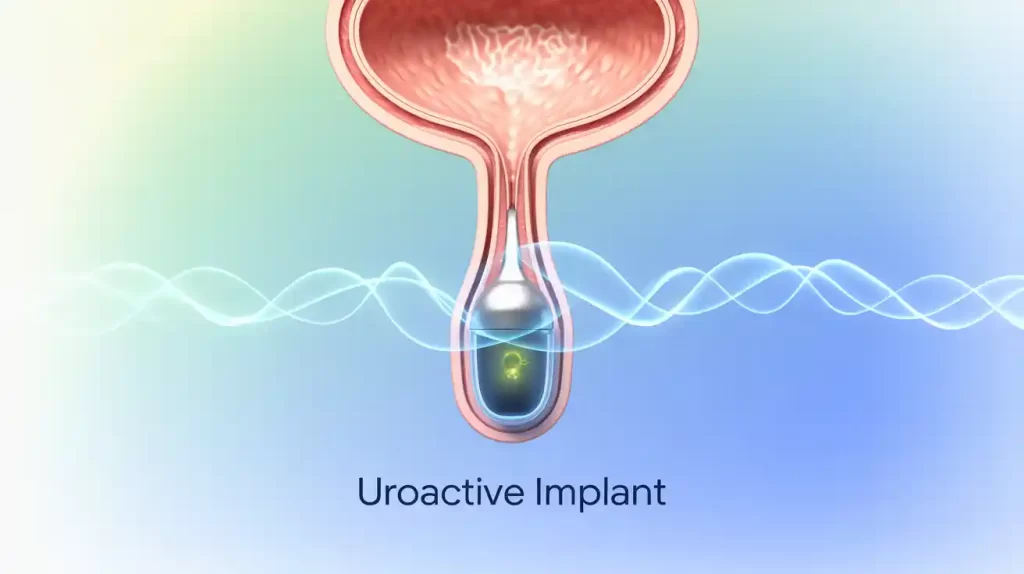
UroMems has received FDA investigational device exemption (IDE) approval to launch its SOPHIA2 clinical trial evaluating the UroActive smart implant for stress urinary incontinence (SUI) in men. The French National Agency for the Safety of Medicines and Health Products (ANSM) has also approved the trial.
The Grenoble-based company’s UroActive implant is an automated artificial urinary sphincter powered by a MyoElectroMechanical system (MEMS). It autonomously adjusts to patient activity, eliminating the need for manual control and aiming to improve ease of use and quality of life. The implant is placed in the urethral duct in men and bladder neck in women.
Following encouraging early feasibility data and the first female implant last year, SOPHIA2 aims to assess safety and efficacy, with results forming the basis for regulatory submissions in the U.S. and Europe.
CEO Hamid Lamraoui called this a major milestone after more than a decade of development, heralding a new era for SUI treatment.
Follow MEDWIRE.AI for innovations in urological devices.