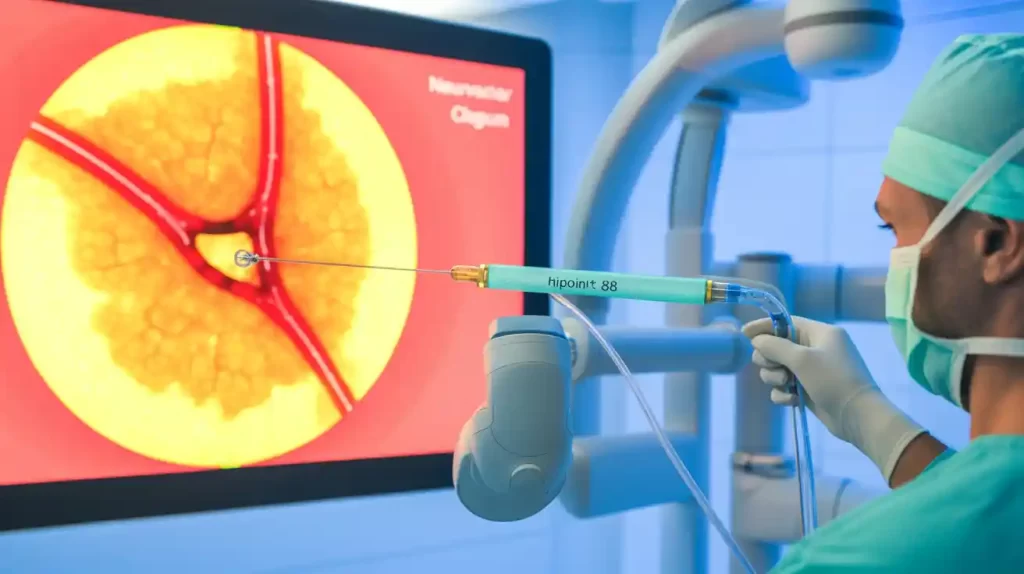
Route 92 Medical reported positive outcomes from a retrospective multicenter study of its FDA-cleared HiPoint 88 reperfusion system for acute ischemic stroke. The HiPoint 88 catheter, featuring the Tenzing 8 Monopoint approach, is the first FDA-cleared 0.088” catheter for direct aspiration of large vessel occlusions.
Data from 193 patients showed a 96.2% successful delivery rate to the target occlusion. The study met its primary endpoints with a first-pass effect of mTICI 2c/3 reperfusion in 57.5% of cases and mTICI 2b/2c/3 in 68.4%. These results demonstrate effective clot removal on the initial aspiration attempt.
Route 92 also announced the upcoming SUMMIT RISE study, a prospective 500-patient trial to further evaluate the safety and efficacy of its neurovascular intervention portfolio.
“We continue to build evidence supporting the Tenzing-based approach’s ability to safely reach and aspirate clots while improving first-pass success rates,” said CEO Dr. Tony Chou.
Follow MEDWIRE.AI for stroke intervention updates.