GT Medical Technologies closed an oversubscribed $53 million Series D funding round to accelerate U.S. commercial expansion and clinical research for its GammaTile brain tumor treatment.
The round included $16 million from new investors such as FemHealth Ventures and Warren Point Capital, supplementing an earlier $37 million close led by Evidity Health Capital and others.
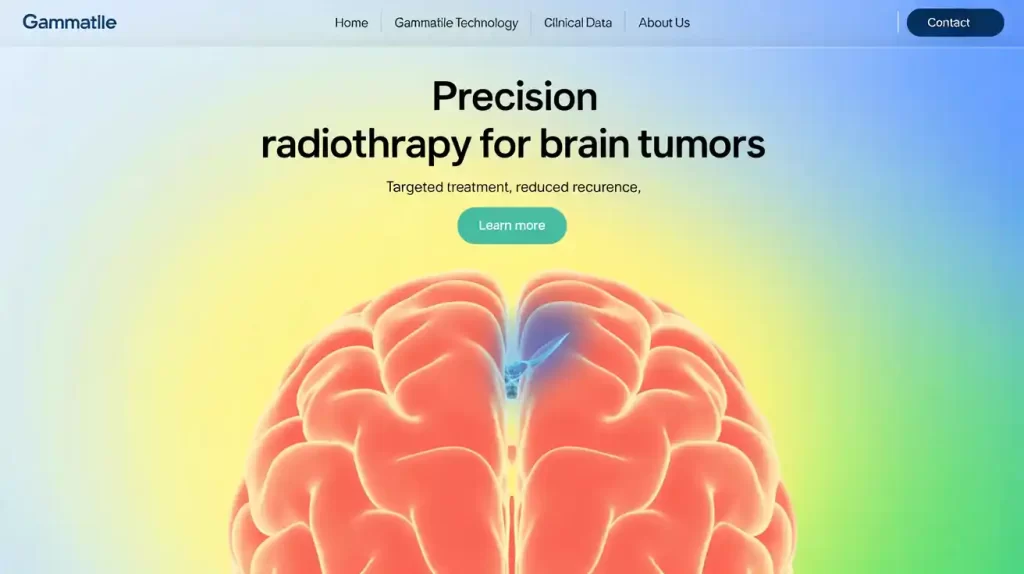
GammaTile is an FDA-cleared, bioresorbable radiotherapy implant delivering localized Cesium-131 radiation directly to tumor cavities to reduce recurrence while sparing healthy tissue. GT Medical plans to complete enrollment in a trial for brain metastases and initiate a study for newly diagnosed glioblastoma patients.
CEO Per Langoe emphasized the funding’s role in sustaining patient-focused growth and innovation.
Follow MEDWIRE.AI for oncology and brain tumor treatment updates.