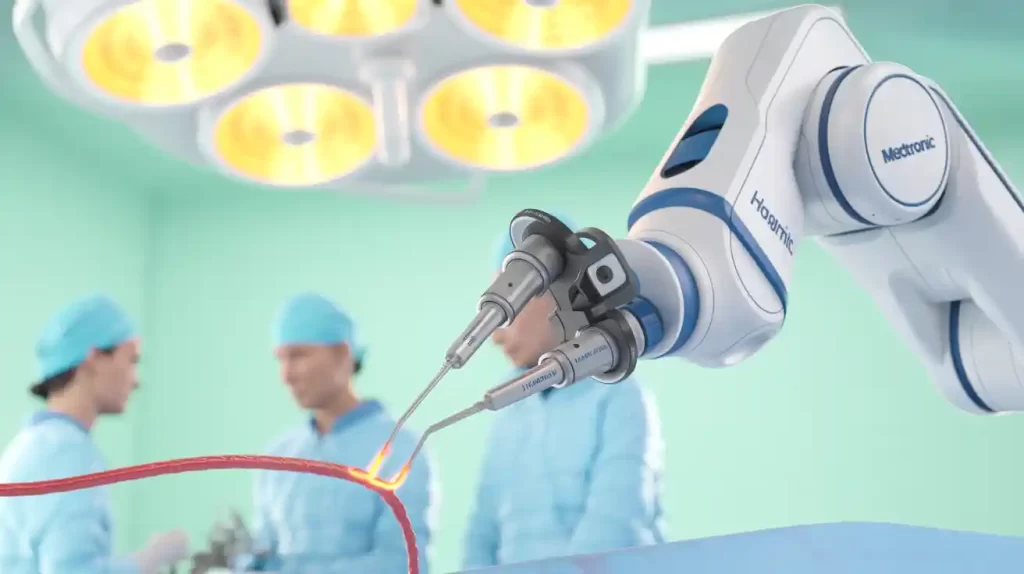
Medtronic has secured CE mark approval for its LigaSure vessel sealing instrument for use with the Hugo robotic-assisted surgery (RAS) system. The LigaSure RAS Maryland device, powered by the Valleylab FT10 energy platform, expands Hugo’s capabilities for gynecologic, general, and urologic procedures in Europe.
This marks the first robotic-driven LigaSure instrument, enhancing vessel sealing and cutting of vessels up to 7 mm with minimal thermal spread. Hugo now operates in 30 countries across five continents, with FDA review ongoing for a U.S. urology indication.
Follow MEDWIRE.AI for surgical robotics market updates.