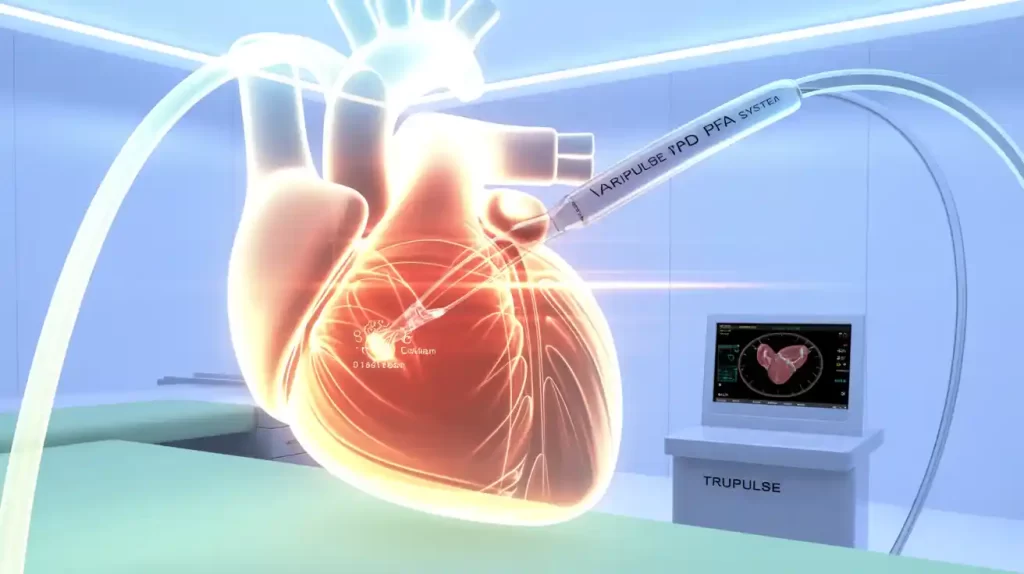
Johnson & Johnson MedTech (NYSE: JNJ) announced FDA approval for an updated irrigation flow rate on its Varipulse pulsed field ablation (PFA) platform, reflecting refinements based on real-world clinical experience.
Varipulse combines PFA technology with the Carto 3 3D cardiac mapping system and features a variable-loop, multi-electrode catheter built from laser-cut nitinol tubing. The platform also includes the TruPulse generator.
Originally cleared in November 2024, Varipulse faced a brief U.S. sales pause early this year after four neurovascular events but resumed after review. Since launch, over 10,000 procedures have been performed globally, with a neurovascular adverse event rate under 0.5%.
Michael Bodner, group chair of Electrophysiology & Neurovascular, highlighted the update as part of Johnson & Johnson’s commitment to iterative product improvement and procedural confidence.
Follow MEDWIRE.AI for updates on cardiac ablation and electrophysiology technologies.