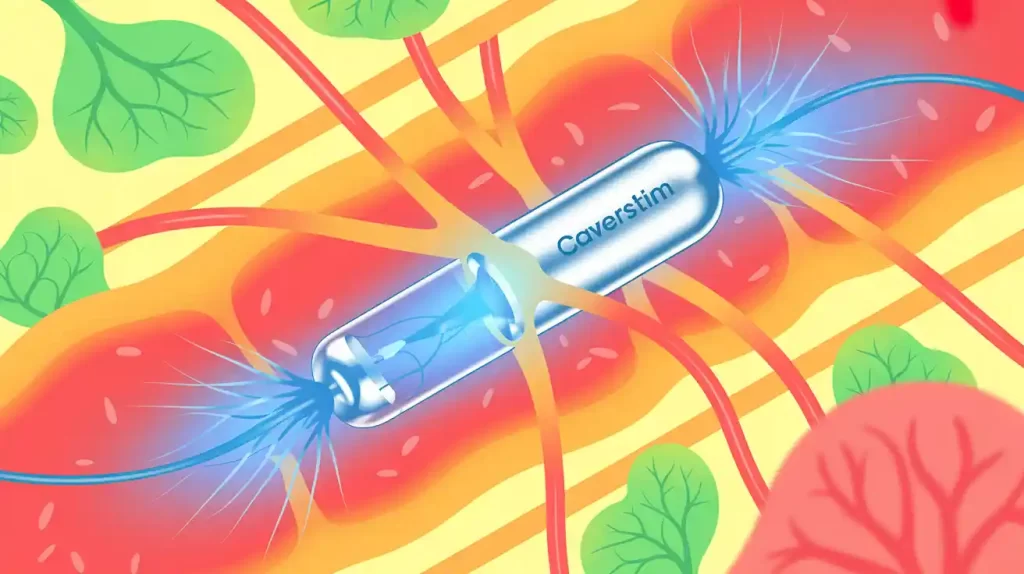
Comphya closed an oversubscribed Series A round totaling CHF 7.5 million (~$8.4 million) to support clinical development of CaverSTIM, its implantable neurostimulation system targeting erectile dysfunction (ED) unresponsive to medication.
The funding will enable a U.S. pilot study under FDA investigational device exemption, with Johns Hopkins University as the first active site, and prepare for a pivotal U.S. trial planned for 2026. It will also expand ongoing Australian studies demonstrating positive safety and efficacy signals.
CaverSTIM stimulates cavernous nerves via a rechargeable implant with dual electrode arrays, aiming to restore natural erections, particularly in men post-prostate cancer treatment. CEO Pim van Wesel noted the financing as a key step toward bringing a transformative solution to patients.
Follow MEDWIRE.AI for the latest on neurostimulation and urological innovations.