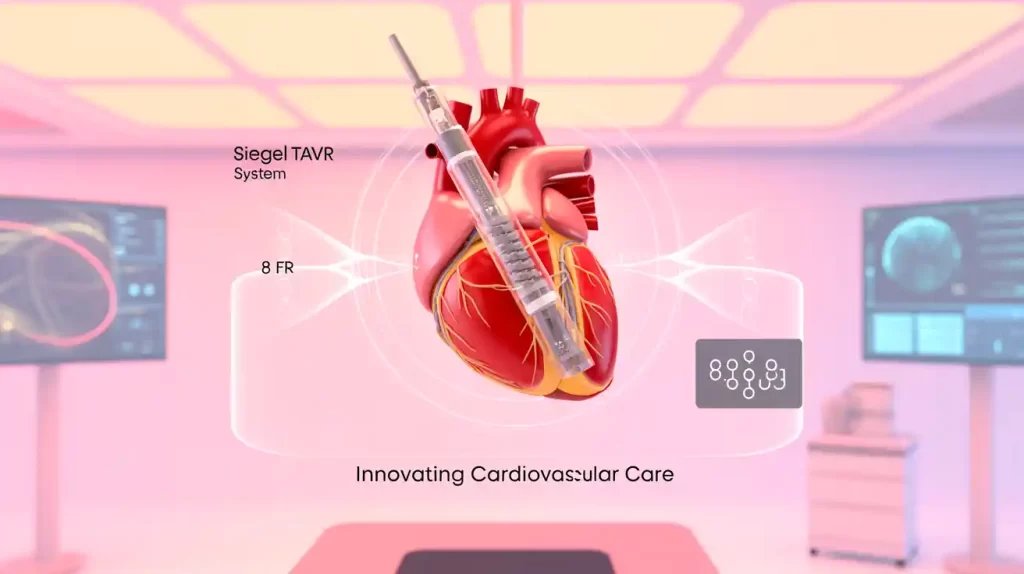
MiRus announced encouraging 30-day results from its U.S. early feasibility study (EFS) of the Siegel transcatheter aortic valve replacement (TAVR) system. Involving 15 patients with severe aortic stenosis at five sites, the study reported zero deaths, strokes, rehospitalizations, vascular complications, paravalvular leaks, or permanent pacemaker implants.
The Siegel system features an 8 Fr delivery sheath enabling less invasive procedures and wider patient access, especially for women. It is the only nickel-free TAVR device, benefiting patients with nickel allergies. Designed with Rhenium alloys, Siegel offers high strength, fatigue resistance, and no foreshortening during implantation, enhancing precise delivery.
“This TAVR’s unique design should reduce pacemaker and vascular complications,” said lead investigator Dr. Pradeep Yadav.
Follow MEDWIRE.AI for updates on TAVR innovation and clinical trials.