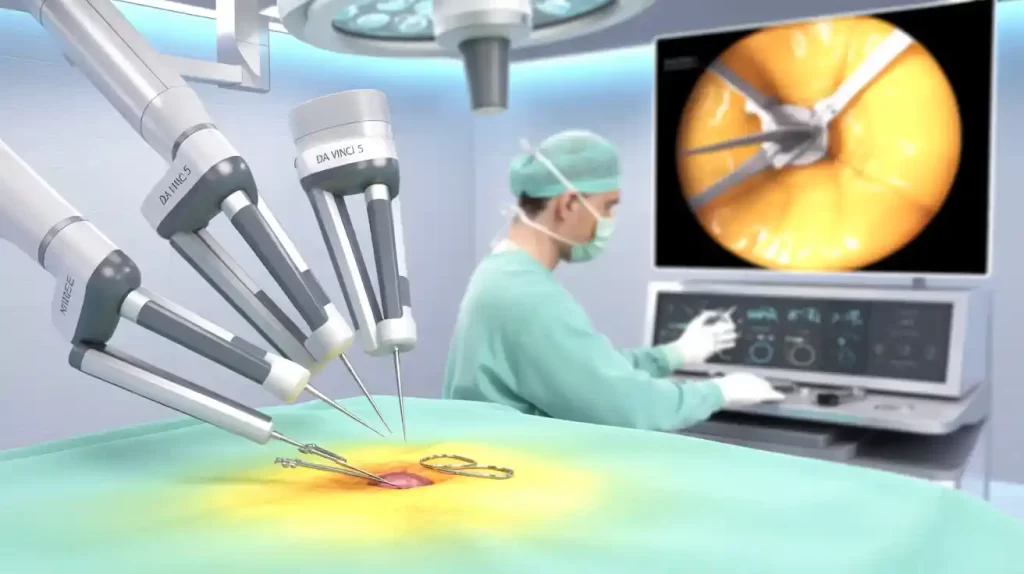
Intuitive Surgical announced it received CE mark approval for its latest da Vinci 5 surgical robotic system, clearing the way for use across Europe in adult and pediatric minimally invasive surgeries. The system supports procedures in the abdominopelvic area, including urologic, gynecologic, general laparoscopic, and thoracoscopic surgeries.
The da Vinci 5 features over 150 enhancements, including force feedback technology, improved 3D vision, advanced tremor and vibration control, and an ergonomic surgeon console. The system received FDA clearance last year and began its U.S. commercial rollout earlier in 2025.
“We’re proud to bring da Vinci 5 to European healthcare systems, offering advanced capabilities that improve outcomes, efficiency, and surgical insights,” said CEO Dave Rosa.
Follow MEDWIRE.AI for updates on surgical robotics and innovation.