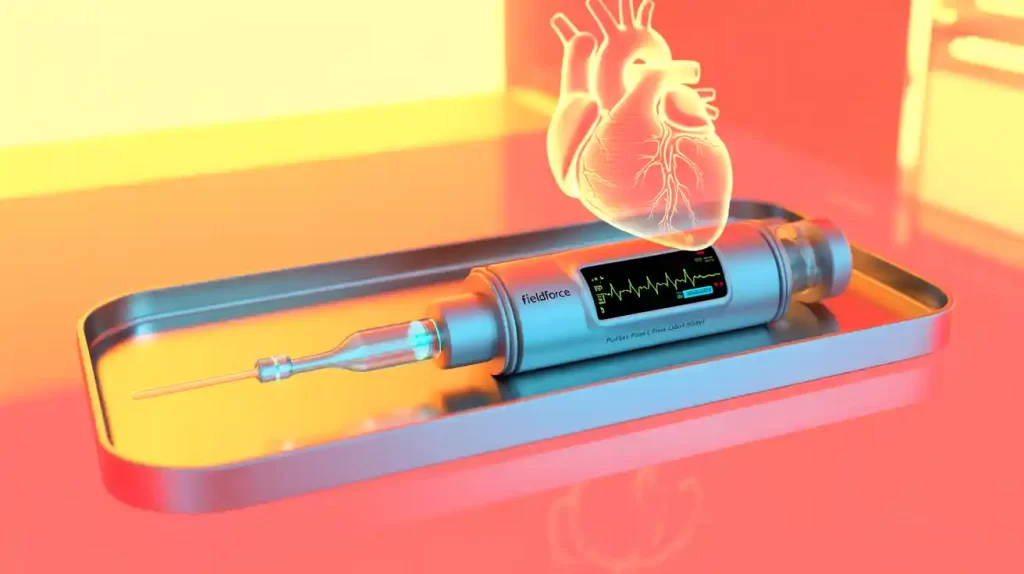
Field Medical announced it closed a $35 million Series B funding round led by BioStar Capital and Cue Growth, boosting its total funding to $75 million.
The Cardiff-by-the-Sea, California-based company plans to use the proceeds to support a pivotal trial for its FieldForce pulsed field ablation (PFA) system, designed as the first contact force, focal PFA catheter with an instantaneous workflow.
FieldForce received FDA breakthrough device designation in December 2024. Unlike other PFA platforms focusing on atrial fibrillation, Field Medical targets ventricular tachycardia (VT), an underpenetrated area in electrophysiology.
“Ventricular tachycardia is among the most underpenetrated segments in electrophysiology, yet physicians still lack the tools they need,” said CEO Dr. Steven Mickelsen. “We are poised to unlock this opportunity with next-generation therapy built for speed, precision and real clinical progress.”
Follow MEDWIRE.AI for electrophysiology and cardiac device updates.