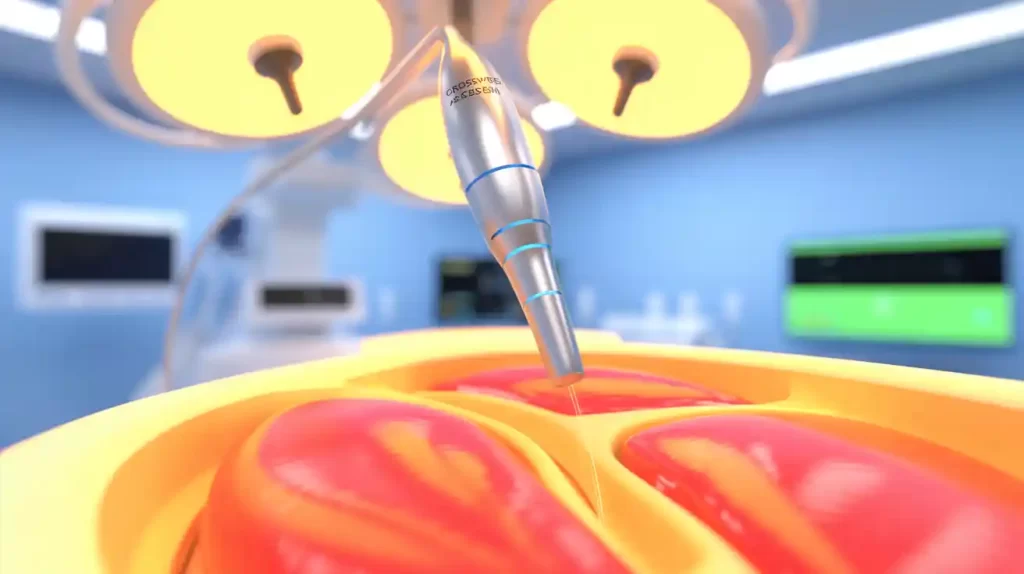
June 27, 2025 — Circa Scientific announced the successful completion of the limited market release of its CrossWise RF transseptal access system, designed to enhance precision and control during left atrial access procedures.
The Colorado-based company’s zero-exchange RF system delivers energy via a specialized cannula that maintains shape and stability during septal crossing, minimizing unintended movement even in challenging anatomies. Bench testing showed clean, rapid punctures—some as fast as 290 milliseconds—across varied septal tissues, including thickened and fibrotic.
Cleared by the FDA in September 2024, CrossWise eliminates wire or sheath exchanges to streamline procedures while maintaining positioning. Following early commercial use starting January 2025, Circa plans to expand U.S. commercial availability soon.
CEO Lee Geist emphasized physician feedback highlighting improved control and versatility with CrossWise’s sheath compatibility and cannula-first design.
Follow MEDWIRE.AI for interventional cardiology device updates.