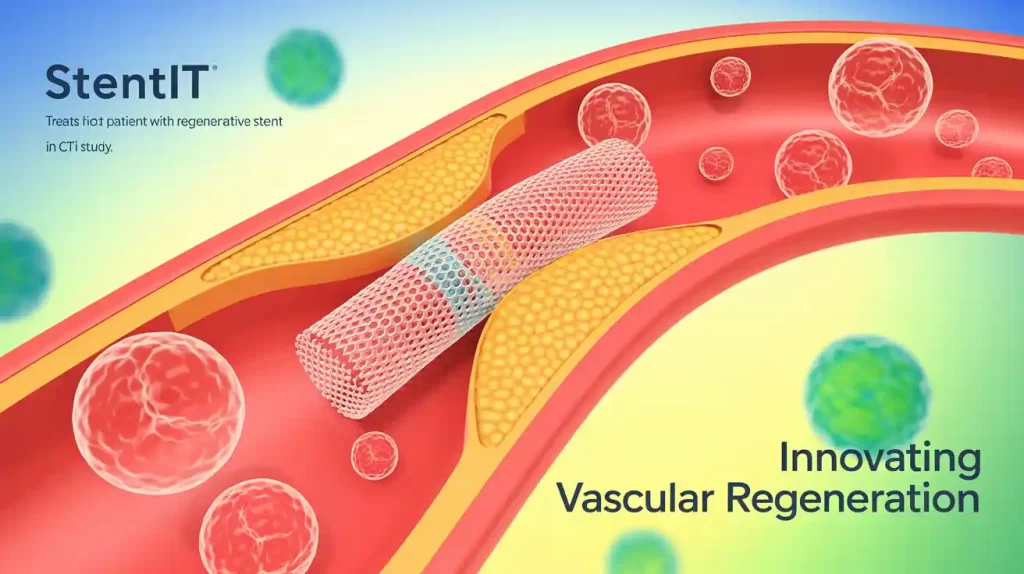
STENTiT has successfully implanted its Resorbable Fibrillated Scaffold in the first patient enrolled in its VITAL-IT 1 clinical study for chronic limb-threatening ischemia (CLTI) below the knee.
The prospective, non-randomized study, conducted at the Medical University of Graz in Austria, will enroll up to 10 patients and follow them for 24 months. The bioresorbable scaffold provides immediate structural support while its porous microfiber mesh guides the patient’s own cells to regenerate new vascular tissue. Over time, the scaffold dissolves naturally, leaving behind a restored artery.
“This marks a major milestone for regenerative vascular therapies,” said CEO Bart Sanders. “We aim to help the body rebuild arteries and reduce the need for repeat procedures.”
CLTI is the most advanced stage of peripheral artery disease, affecting 3.5 million people in Europe and the U.S., with over 250,000 amputations annually.
“This first-in-human study will show whether this novel approach can improve long-term outcomes for CLTI patients,” said Prof. Marianne Brodmann, principal investigator.
Follow MEDWIRE.AI for more updates on regenerative vascular innovations.