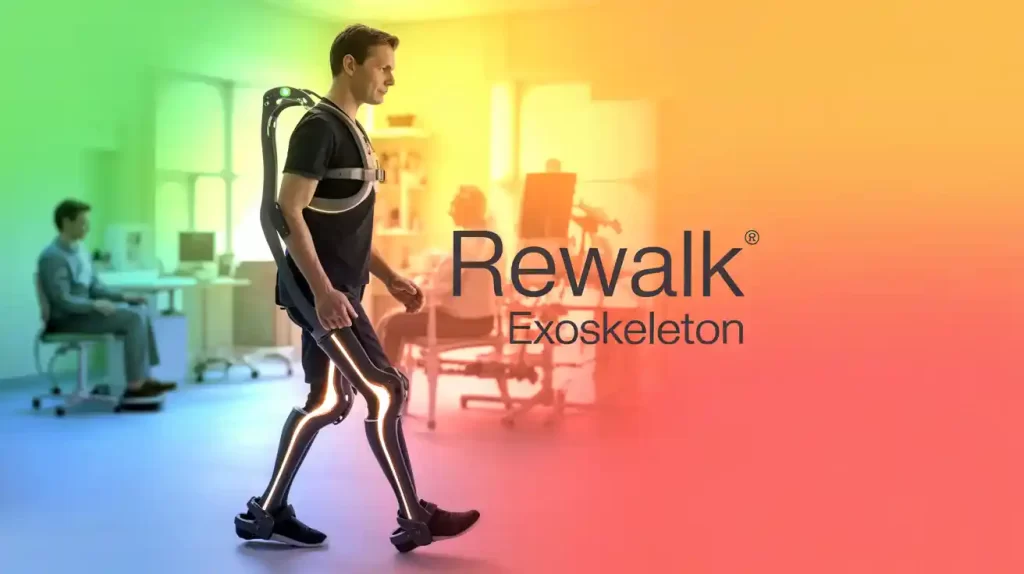
In a significant decision for spinal cord injury care, a U.S. Administrative Law Judge (ALJ) has ruled that Lifeward’s ReWalk Personal Exoskeleton qualifies for Medicare coverage as a “reasonable and necessary” medical device.
The ruling was made in response to an appeal by a Medicare beneficiary, supporting ReWalk’s use as a clinically backed intervention that enables ambulation for individuals with spinal cord injuries (SCI). It builds on momentum from CMS’s 2023 reimbursement policy and its finalized pathway in 2024.
Key Highlights:
- ReWalk 7, Lifeward’s next-gen personal exoskeleton, launched earlier this year in the U.S.
- Lifeward says the ruling sets a legal precedent that may lead to expanded commercial payer coverage.
- The company is also raising $2.6 million through a public offering to support commercialization and operations.
“This ruling not only clarifies Medicare access but also helps open doors for broader adoption,” said Lifeward CEO Mark Grant.
Follow MEDWIRE.AI for updates on MedTech coverage and breakthrough mobility technologies.