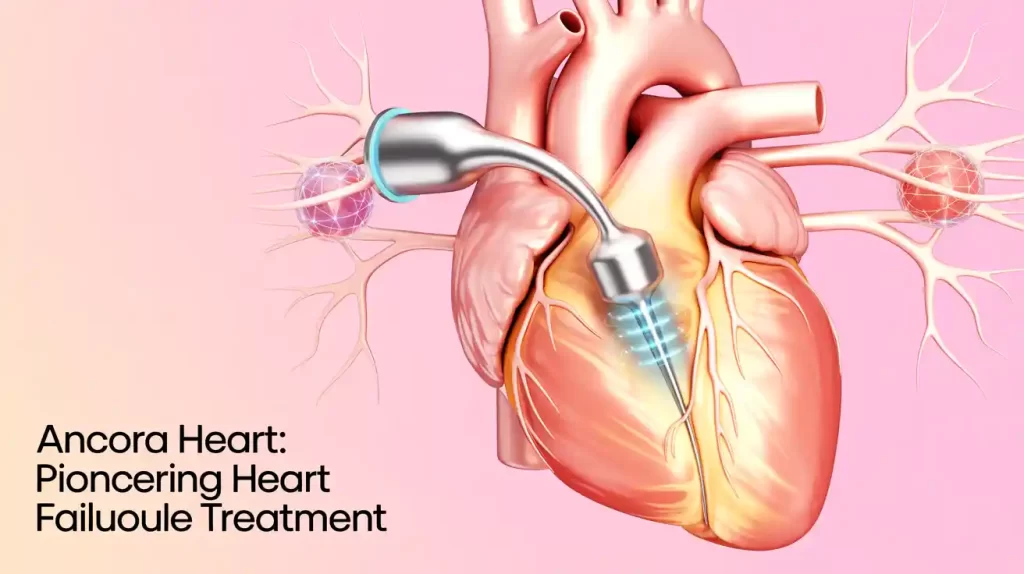
Ancora Heart has announced the enrollment of 250 patients in its CORCINCH-HF pivotal trial, evaluating the AccuCinch Transcatheter Left Ventricular Restoration System for heart failure patients with reduced ejection fraction (HFrEF). This milestone supports the company’s plan to submit premarket approval (PMA) data to the FDA, with enrollment continuing toward a total of 400 participants.
The AccuCinch system is a catheter-based device designed to reduce left ventricular size, decrease wall stress, and support heart structure in patients who have not responded sufficiently to guideline-directed medical therapy and are not candidates for ventricular assist devices or heart transplantation. It received FDA breakthrough device designation in 2022.
Jeff Closs, President and CEO of Ancora Heart, highlighted that the AccuCinch System is the only fully transcatheter procedure aimed at treating an enlarged left ventricle. He praised the milestone as a key accomplishment in heart failure research and a testament to collaborative efforts across trial sites.
The CORCINCH-HF trial is a prospective, randomized, multicenter study enrolling patients worldwide. Initial safety and effectiveness data will be analyzed after six months for the first 250 patients, with final results expected after all 400 participants complete 12 months.
Dr. Ulrich Jorde, global co-principal investigator, noted that despite medical advances, many heart failure patients still suffer debilitating symptoms. He described the milestone as a crucial step toward evaluating the potential of this treatment to improve patient longevity and quality of life.
Follow MEDWIRE.AI for the latest updates in heart failure therapies.