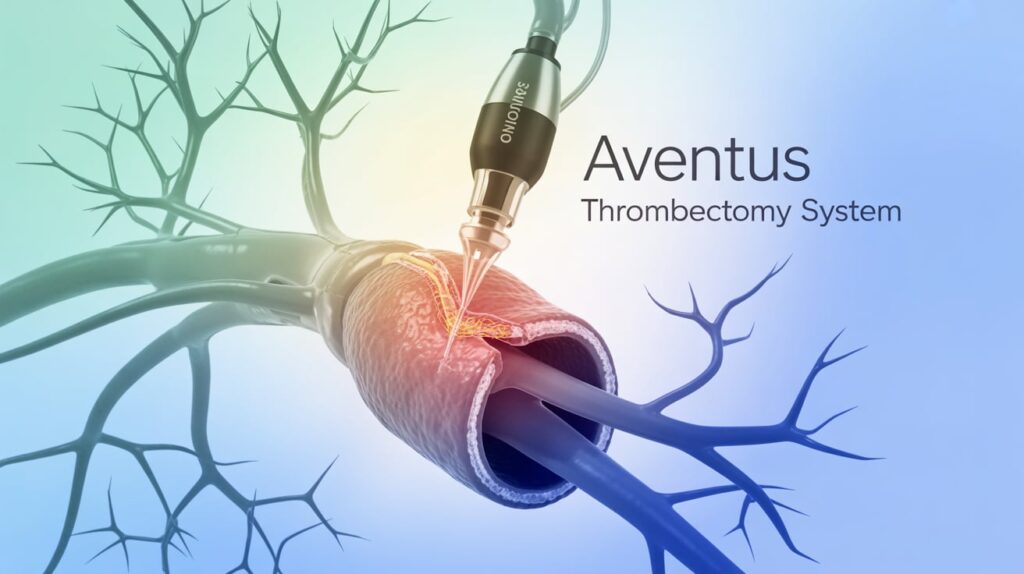
Inquis Medical announced the FDA has granted expanded 510(k) clearance for its Aventus thrombectomy system to treat pulmonary embolism (PE). This follows positive study results demonstrating safety and efficacy in acute intermediate-risk PE patients.
The Aventus platform features proprietary tissue-sensing technology, providing real-time feedback on tissue composition for precise clot removal. It aims to overcome limitations of existing thrombectomy devices by combining clot extraction with streamlined blood return in one integrated system.
Last year, Aventus received FDA clearance for peripheral indications, supported by a $40 million Series B funding round in late 2024.
Co-CEO Mojgan Saadat highlighted the platform’s precision and procedural efficiency, calling the rapid clearance a testament to strong clinical data and the Inquis team’s efforts.
Follow MEDWIRE.AI for updates on vascular intervention innovations.