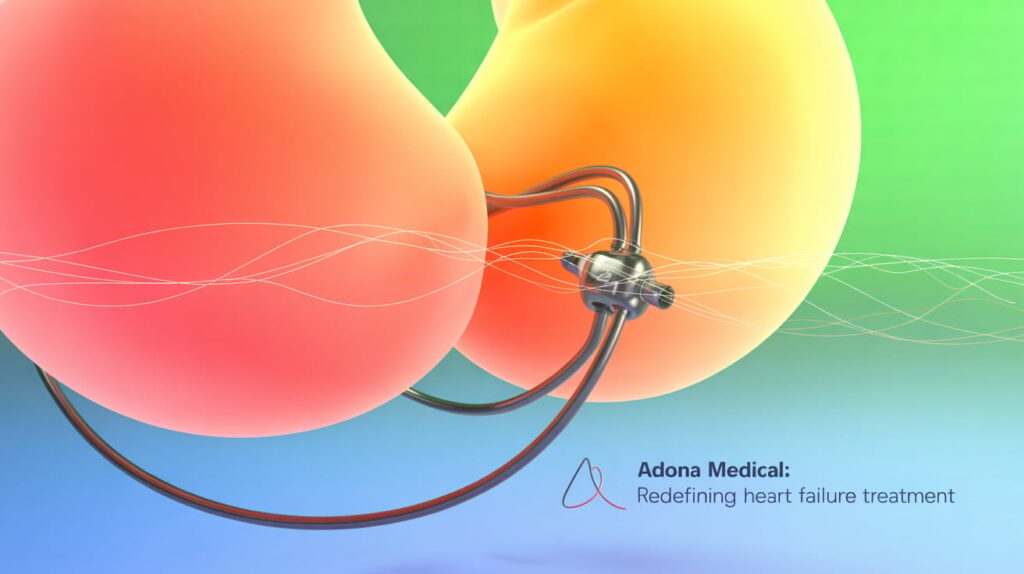
Adona Medical has completed enrollment of 10 patients in its ATHENS-HF first-in-human study of a novel adjustable interatrial shunt with integrated bi-atrial pressure sensors.
The nitinol device allows post-implant adjustment of the flow channel size to tailor shunting based on patient needs over time. Sensors continuously measure left and right atrial pressures without patient interaction.
The trial includes heart failure patients with both preserved and reduced ejection fraction. Implantation success stands at 100% with no major procedural complications or significant cardiovascular or neurovascular adverse events reported. Most patients reached the 90-day follow-up and underwent shunt size adaptations as planned.
CEO Brian Fahey calls this a pivotal milestone and plans to advance the clinical program with next-generation devices later this year.
Follow MEDWIRE.AI for updates on heart failure device innovation.