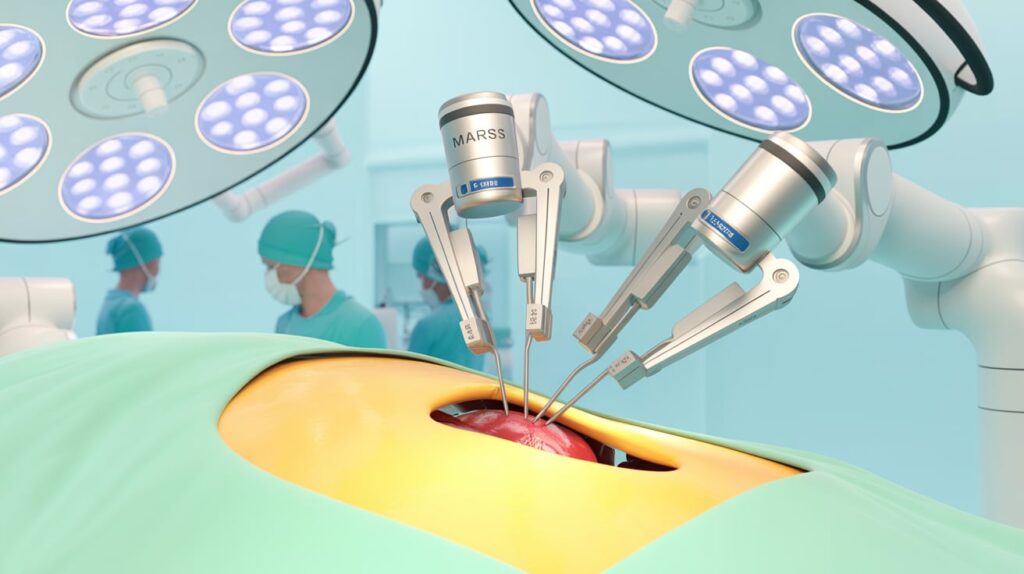
Levita Magnetics has received expanded FDA 510(k) clearance for its MARS system to include bariatric and hiatal hernia surgeries.
MARS uses magnetic-assisted technology to retract organs and enhance visibility with fewer incisions. The clearance also covers a new 12.5 mm magnetic grasper, designed for high-BMI patients and improved liver control during surgery.
The compact system builds on Levita’s earlier magnetic surgical tech, offering a less invasive and more controlled robotic solution.
Follow MEDWIRE.AI for the latest in surgical robotics innovation.