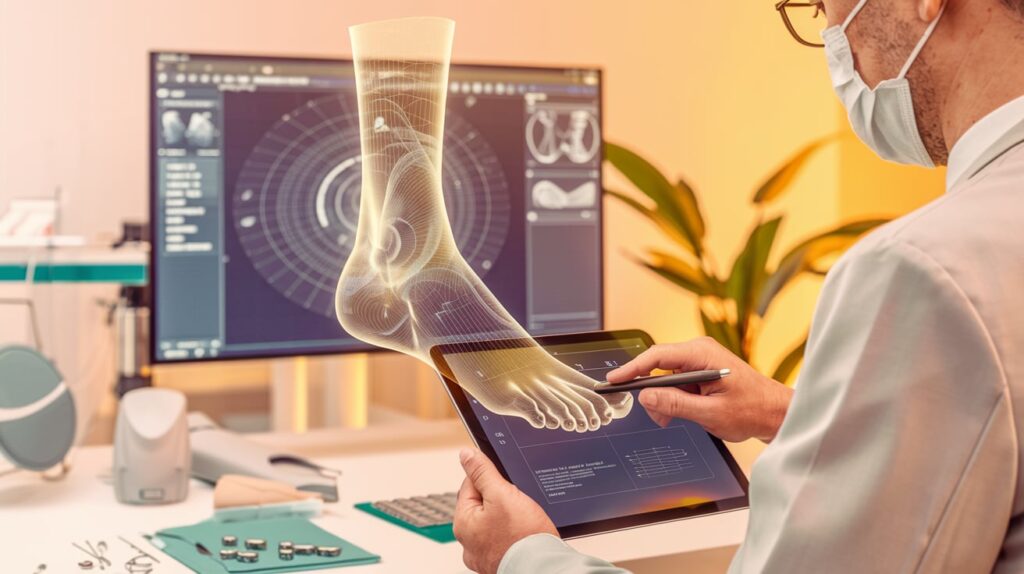
MedCAD, based in Dallas, has received 510(k) clearance for its AccuStride Foot and Ankle System, a patient-specific precision device designed to assist surgeons in treating multiple related pathologies in the foot and ankle in a single procedure. This marks MedCAD’s first entry into the lower extremities market, complementing its established reputation in custom-designed implants and surgical devices.
The AccuStride System leverages MedCAD’s proprietary software to offer surgeons a customizable solution for complex cases, such as proximal phalanx and metatarsal arthroplasty, Lapidus revisions, and cavus foot corrections. The system’s components are constructed from UV-curable acrylate polymers or titanium alloys, allowing delivery in as little as five days after receiving medical imaging and surgeon design approval.
MedCAD’s CEO, Nancy Hairston, highlighted the system’s potential to reduce surgery duration and frequency while delivering durable outcomes. The company’s focus on patient-matched solutions aims to provide surgeons with anatomically tailored options that improve patient care and produce life-changing results.
The AccuStride System is intended for patients aged 12 and older, offering surgeons advanced tools for more efficient, effective foot and ankle surgeries.
Follow MEDWIRE.AI for more updates on innovations in medical devices.